Direct observation of preferential processing of clustered abasic DNAdamages with APE1 in TATA box and CpG island by reaction kinetics and fluorescence dynamics
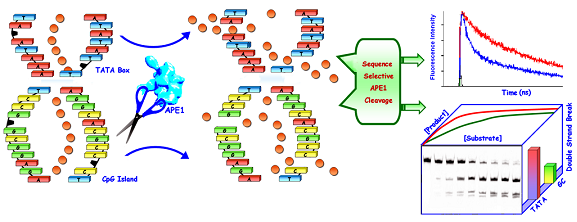
Sequences like the core element of TATA box and CpG island are frequently encountered in the genome and related to transcription. The fate of repair of clustered abasic sites in such sequences of genomic importance is largely unknown. This prompted us to investigate the sequence dependence of cleavage efficiency of APE1 enzyme at abasic sites within the core sequences of TATA box and CpG island using fluorescence dynamics and reaction kinetics. Simultaneous molecular dynamics study through steady state and time resolved fluorescence spectroscopy using unique ethidium bromide dye release assay confirmed an elevated amount of abasic site cleavage of the TATA box sequence as compared to the coreCpG island. A greater binding effect of APE1 enzyme on TATA sequence was observed that signifies a prominent role of the sequence context of theDNA substrate. A faster response from APE1 was obtained for clustered abasic damage repair of TATA box core sequences than CpG island consensus sequences. The repair refractivity of the bistranded clustered abasic sites arise from the slow processing of the second abasic site, consequently resulting in decreased overall production of potentially lethal double strand breaks. This is reported in the journal Mutation Research/Fundamental and Molecular Mechanisms of Mutagenesis (2014, 766–767, 56–65) by Dr. Prolay Das and Dr. Debabrata Seth and their research groups.